
Articles on Food and Drug Administration (FDA)
Displaying 61 - 80 of 163 articles

A new study suggests the market alone will not deter or punish pharmaceutical companies whose products turn out to have adverse effects after they have been approved.

When making the decision whether to vaccinate children aged five to 11 against COVID-19, regulators in Canada must rely on sound ethics as well as sound science.

Regulators are currently reviewing the safety and efficacy data of the Pfizer vaccine for five to 11 year olds before deciding whether to approve its use in this age group.

Three tobacco-flavored cartridges and a vaping device have been approved by the FDA for sale in the US. It comes after a decadelong debate over e-cigarettes.

In light of mounting research showing the serious risks of contracting COVID-19 during pregnancy, the CDC is re-upping its urgency that pregnant women get their shots.

The FDA has chosen not to regulate lab-developed tests like the ones at the center of the Theranos trial.
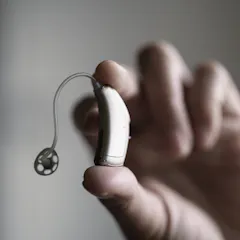
Only 3.7% of people in the U.S. with hearing difficulty own hearing aids. Thanks to a federal law in progress of being implemented, OTC hearing aids may help bridge the gap.

An FDA panel has voted against recommending approval of a booster COVID-19 shot for the general population – disappointing some public health officials.

Subtly shifting the crafting and delivery of public health messaging on COVID-19 vaccines could go a long way toward persuading many of the unvaccinated to get the shot.

Making rapid antigen tests for COVID-19 cheaper and more accessible can catch the infectious cases before they spread and help everyone resume normal activities safely.

An infectious disease doctor explains the science behind COVID-19 vaccines at a level that children – and adults – of all ages can understand.

The U.S. FDA has approved the first COVID-19 vaccine. How is approval different from emergency use authorization, and what difference will it make to a vaccine that’s already in global use?

The FDA has largely lost its ability to regulate the myriad pills, powders and potions that promise to grow muscle, shed body fat and improve your focus.

While emergency use authorization gets vaccines out to the public more quickly, it doesn’t skip any steps that full approval requires.

Researchers are testing an antibody shot to protect people from Lyme disease-causing bacteria.

Ongoing tracking is meant to spot very rare risks – like the connection between the Johnson & Johnson shot and Guillain-Barré syndrome. And it relies on public reporting.

COVID-19 has exacerbated a backlog of domestic and foreign drug manufacturing inspections that the FDA is still too short-staffed to adequately deal with.

The new drug is based on the idea that a build-up of amyloid in the brain leads to the disease. But that hypothesis has been under scrutiny lately.

The FDA approved Alzheimer’s disease drug aducanumab despite minimal evidence of its efficacy. Whether this decision ultimately hurts or helps patients depends on data researchers don’t yet have.

The FDA receives almost half its funding from companies it regulates, such as drug and medical device makers. Is this something you should be concerned about?





